Stephen Aldehyde Synthesis, Mechanism and Application
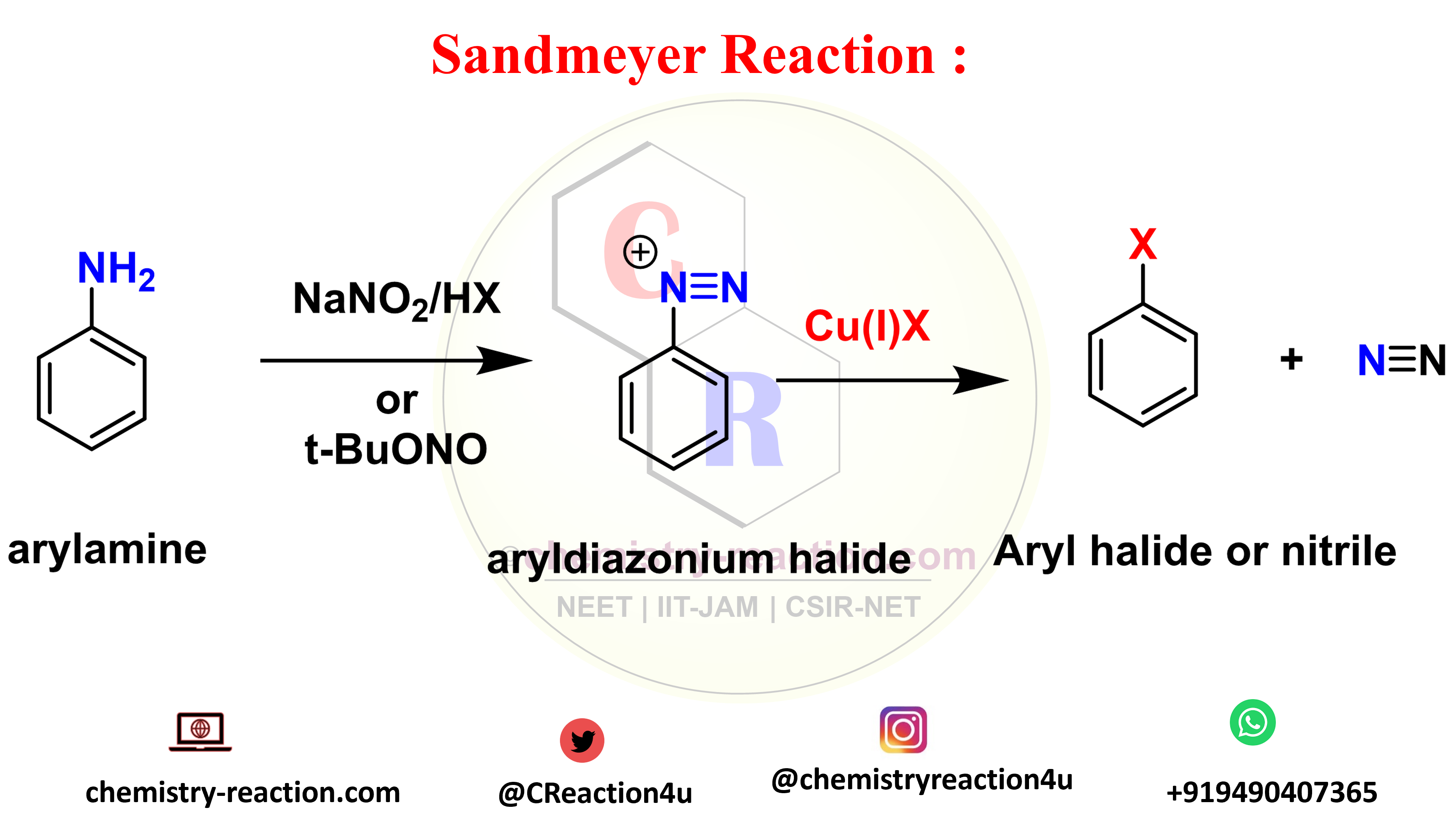
Stephen Reduction Reaction is also known as Stephen aldehyde synthesis or Stephen reaction in organic chemistry. here is the Stephen Aldehyde Synthesis Mechanism and Application. here aliphatic or aromatic nitrile is converted to imidoyl chloride or uses of sulphuric acid by using hydrochloric acid and further to aliphatic or aromatic aldehyde. Sandmeyer Reaction: Definition| Mechanism| … Read more