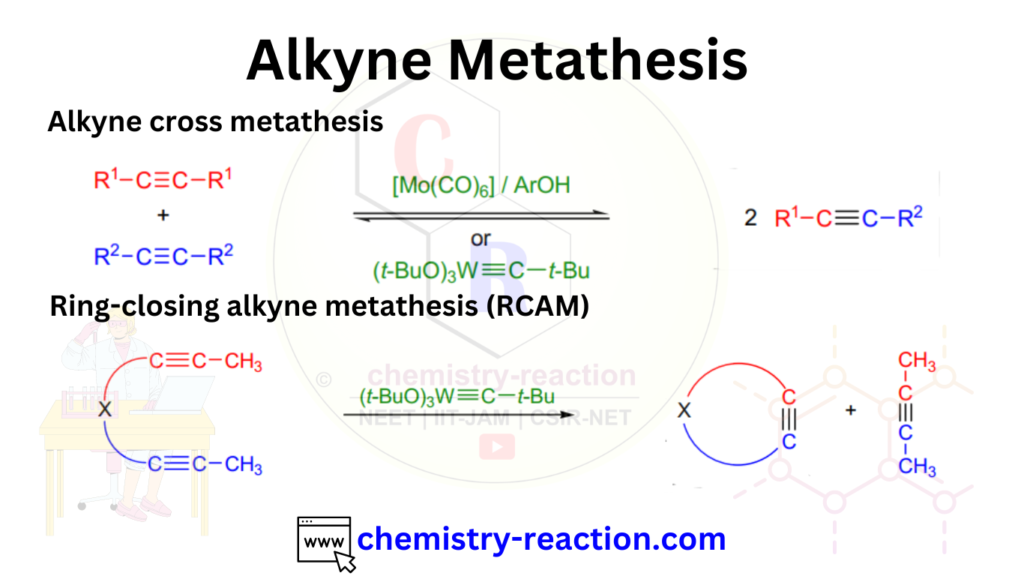
Alkyne metathesis, also known as alkyne cross-metathesis, is a type of metathesis reaction in organic chemistry that involves the exchange of alkynes to form new alkyne products. Metathesis reactions involve the redistribution of alkene or alkyne fragments through the breaking and forming of carbon-carbon double or triple bonds.
In alkyne metathesis, two different alkynes react in the presence of a metal catalyst, typically a transition metal carbene complex. The metal catalyst facilitates the breaking and rearrangement of the carbon-carbon triple bonds, resulting in the formation of new alkyne products.
Alkyne metathesis is a powerful tool in organic synthesis as it enables the selective synthesis of complex alkyne-containing molecules. It allows for the formation of new carbon-carbon triple bonds, which are versatile functional groups in organic chemistry. Alkyne metathesis reactions have been utilized in various applications, including the synthesis of natural products, pharmaceuticals, and materials chemistry. The ability to construct new alkyne bonds efficiently and selectively makes alkyne metathesis a valuable tool in the synthetic toolbox.
Organic Chemistry Reagents Table
Table of Page Contents
Alkyne Metathesis Mechanism:
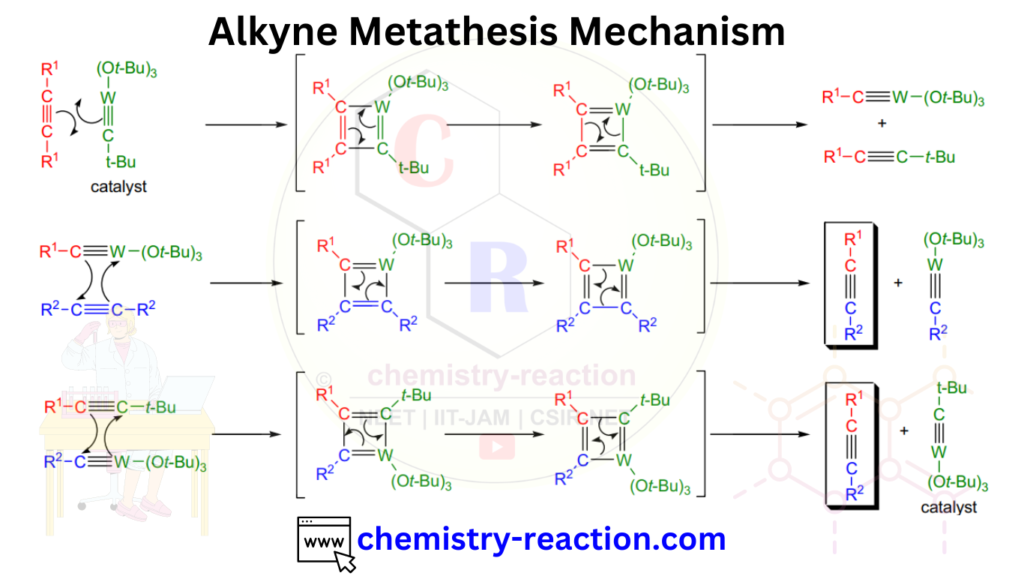
The reaction proceeds through a mechanism similar to other metathesis reactions, involving the formation of metal alkylidene intermediates. The metal catalyst coordinates with the alkyne substrates, leading to the formation of metallacyclobutadiene intermediates. These intermediates undergo rearrangement and reformation of new carbon-carbon triple bonds, resulting in the desired alkyne products.
The mechanism of alkyne metathesis involves a series of steps that lead to the exchange of alkynes and the formation of new alkyne products. Here’s a step-by-step explanation of the mechanism:
- Alkyne coordination:
- Metallacyclobutadiene formation:
- Product formation:
It is important to note that the specific details of the alkyne metathesis mechanism can vary depending on the choice of metal catalyst and reaction conditions. Different metal carbene complexes can exhibit variations in the exact bonding and rearrangement processes. Nevertheless, the general concept of coordination, cyclization, rearrangement, and catalyst regeneration is central to the overall mechanism of alkyne metathesis.
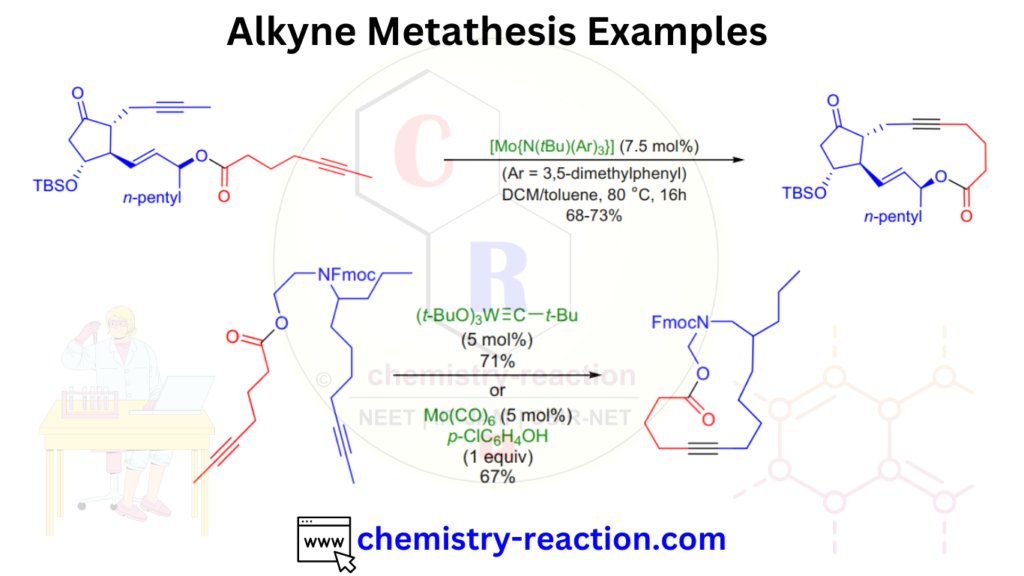
Alkyne Metathesis Examples:
Related Reactions:
- Cross Metathesis
- Alkyne Metathesis Reaction
- Alkyne Cross Metathesis
- RIng-Closing Alkyne Metathesis
References:
- Alkyne Metathesis- sciencedirect
- Alkyne Metathesis in Organic Synthesis – OCP
alkyne metathesis catalyst, Alkyne metathesis reaction formula, Alkyne metathesis reaction ppt, Alkyne metathesis reaction SlideShare, Alkyne metathesis reaction pdf.
- Organic Chemistry Reagents Table
- Sakurai Allylation Reaction Mechanism with Applications
- Saegusa Oxidation Reaction Mechanism with Applications
- Smiles Rearrangement Reaction : Mechanism , Application and Modification
- Snieckus Directed Ortho Metalation Reaction
My name is Pradip Sanjay W. I’m an organic chemist originally from Maharashtra, India. I have qualified UGC NET-JRF, GATE in chemical sciences and MH-SET exam for assistant professor. I’m currently pursuing my Ph.D. in organic chemistry at the Indian Institute of Technology Hyderabad, India.
