Table of Page Contents
Chugaev Reaction Definition:
The formation of olefins by pyrolysis of the corresponding containing at least one β-hydrogen atom xanthates via cis-elimination is known as the Chugaev elimination reaction. here we will discuses about mechanism and examples of Chugaev Elimination Reaction.
The Chugaev elimination reaction of xanthates is very similar to the ester (acetate) pyrolysis. The by-products (COS, R4-SH) of the Chugaev reaction are very stable, thus making the elimination irreversible. Chugaev Elimination Reaction |mechanism |examples)
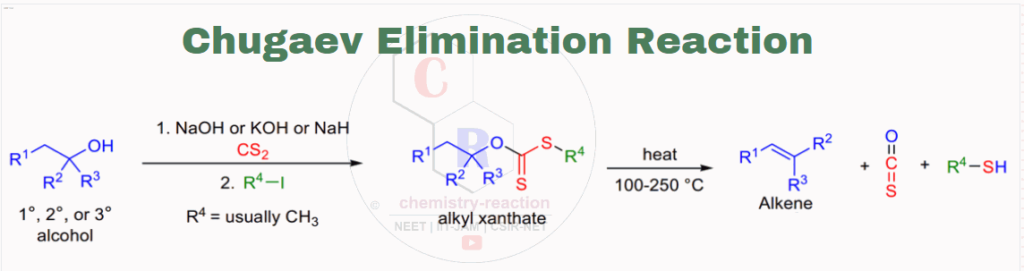
Mechanism of Chugaev Elimination:
The mechanism of Chugaev elimination reaction proceeds through a six-membered transitionstate as described in this picture..
Chugaev reaction is an intramolecular( reaction occurs in same molecule) syn elimination (Ei). Chugaev elimination reaction involve a cis-β-hydrogen atom from the alcohol part and the Thione sulfur atom from the xanthate moiety. Isotopic studies of Chugaev elimination reaction shows involving 34S and 13C showed that the C=S, and not the thiol sulfur atom, closes the ring in the transition state. for the elimination β-hydrogen and the xanthate group should be coplanar mode in the cyclic transition state then only reaction deliver the product.

Chugaev reaction with example:
Chugaev reaction with example are shown in picture.
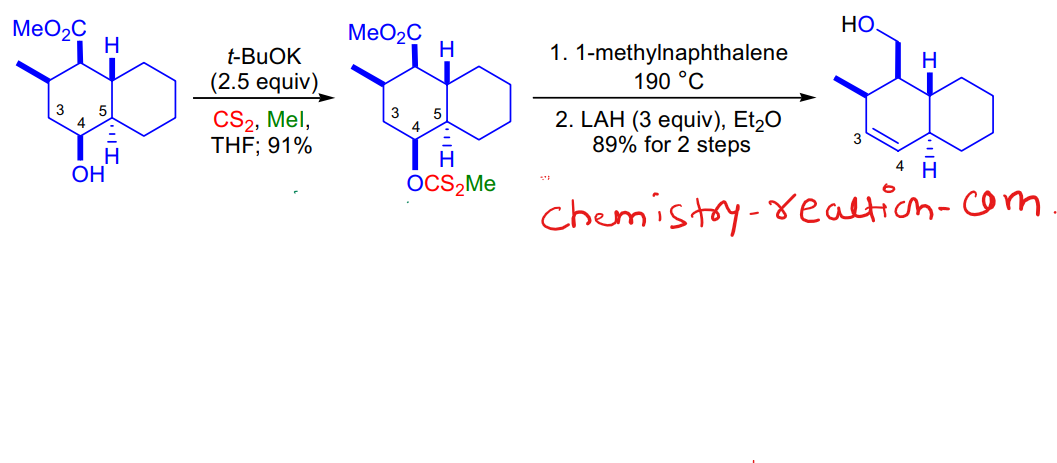
chugaev elimination pdf, chugaev reaction slideshare, chugaev reaction ppt. Chugaev Elimination Reaction |mechanism |examples
Related reaction:
- Burgess dehydration
- Cope rearrangement
- Wharton fragmentation
- Grob fragmentation
- Corey-Winter olefination
- Ramberg-Bäcklund rearrangement
- Hofmann elimination
- Cope elimination
- Barton–McCombie deoxygenation
References:
- sciencedirect.com/chugaev-elimination
- Elimination Reactions. VIII. A trans Chugaev Elimination
My name is Pradip Sanjay W. I’m an organic chemist originally from Maharashtra, India. I have qualified UGC NET-JRF, GATE in chemical sciences and MH-SET exam for assistant professor. I’m currently pursuing my Ph.D. in organic chemistry at the Indian Institute of Technology Hyderabad, India.

1 thought on “Chugaev Elimination Reaction |mechanism |examples”