Table of Page Contents
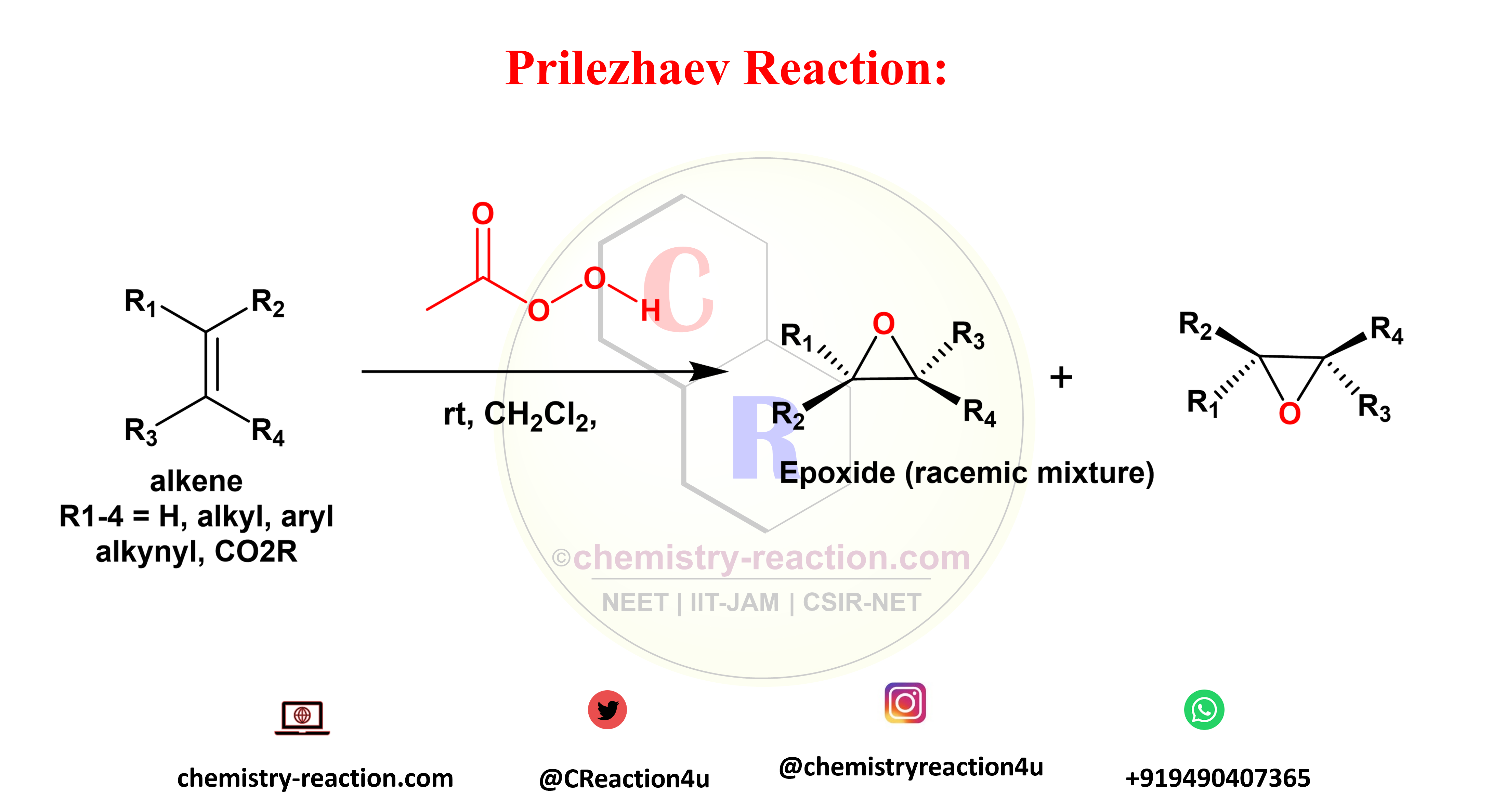
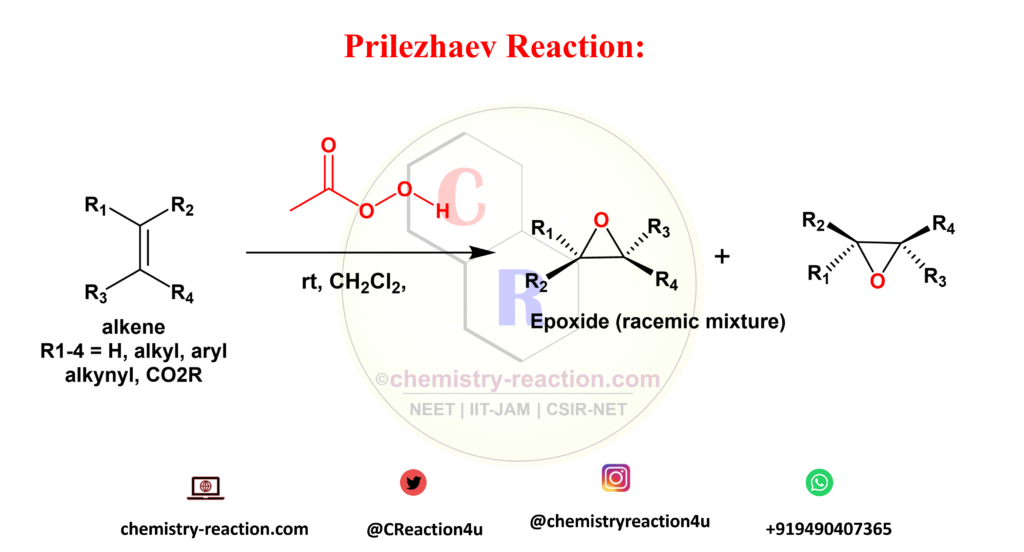
What is Prilezhaev reaction explain ?
Prilezhaev reaction is organic transformation is referred for synthesis of oxiranes (epoxides) from isolated double bonds by using peroxycarboxylic acids as oxidizing agents. This method provides products in racemic mixture form.
Sharpless, Jacobsen, and Shi asymmetric epoxidation is used for synthesis of enantiomerically pure form of oxiranes .
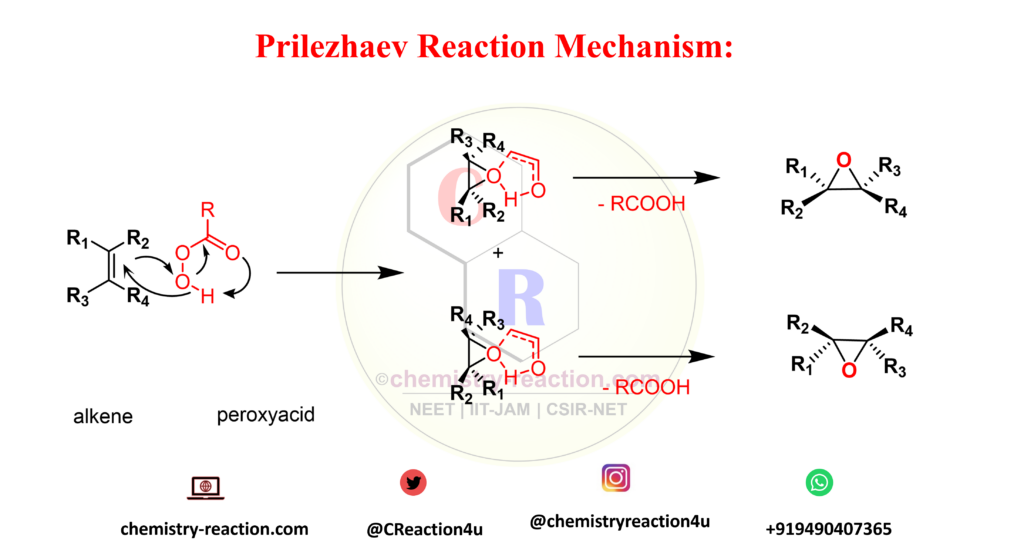
Prilezhaev Reaction Mechanism :
The Prilezhaev reaction is stereospecific, and in all cases a syn addition of the oxygen to the double bond is observed. This observation supports the assumption that the epoxidation of alkenes by peroxyacids is a concerted process. The reaction takes place at the terminal oxygen atom of the peroxyacid, and the π HOMO of the olefin approaches the σ* LUMO of the O-O bond at an angle of 180° (butterfly transition structure).
Related reactions:
References:
- R. Mello, A. Alcalde-Aragonés, M. E. González Núñez, G. Asensio, J. Org. Chem., 2012, 77, 6409-6413.
- N. K. Jana, J. G. Verkade, Org. Lett., 2003, 5, 3787-3790.
- R. S. Porto, M. L. A. A. Vasconcellos, E. Ventura, F. Coelho, Synthesis, 2005, 2297-2306.
My name is Pradip Sanjay W. I’m an organic chemist originally from Maharashtra, India. I have qualified UGC NET-JRF, GATE in chemical sciences and MH-SET exam for assistant professor. I’m currently pursuing my Ph.D. in organic chemistry at the Indian Institute of Technology Hyderabad, India.